Coronavirus and the brain: Diagnosing and treating COVID-19’s neurological effects
A patient is prepared for neurocognitive assessment. (Steven Kim/ Author provided)
BY John F. Connolly
July 23, 2020
As the number of COVID-19 patient records grows, researchers are meticulously combing through the data, searching for a better understanding of the virus and what we can expect in the months and years ahead. What is now of increasing concern in health care is the realization that the virus can not only be severe, but can also have long-term consequences.
Respiratory and skeletal muscle consequences surfaced earlier in the pandemic. More recently, the neurological and neurocognitive aspects of the virus have become a major concern. The neurocognitive symptoms linked to coronaviruses, counting COVID-19, include delirium, both acute and chronic attention and memory deficits linked to hippocampal and cortical damage, as well as learning deficits in both adults and children.
These symptoms feature significantly in a large percentage of COVID-19 patients. In March, a study reported 36.4 per cent of COVID-19 patients have neurological symptoms, including headache, disturbed consciousness and paresthesia (a burning or prickling sensation in parts of the body such as hands, legs and feet). Not surprisingly, severely affected patients are more likely to develop neurological symptoms than patients who have mild or moderate disease.
More recently, a report published in early June in the New England Journal of Medicine reported that 84 per cent of the patient sample had neurologic symptoms. Also, a recent paper in the Lancet using autopsy reports on patients who had severe cases noted brain tissue edema (a life-threatening condition that causes fluid to develop in the brain, causing pressure inside the skull) and neuronal degeneration (when a brain’s neurons break down, lose connectivity and affect brain function).
Assessing neurological symptoms
The percentage of COVID-19 patients with neurological symptoms varies greatly between studies. One of the reasons is inconsistent assessment methods. Research has shown behavioural assessment (testing based on things like the patient’s visual responses, motor function and communication) is frequently inaccurate.

Reliable neurocognitive assessment procedures will be essential both in accurately assessing active and post-COVID patients’ cognitive abilities, and in tracking their recovery. Objective assessments of brain function can help pinpoint when neurocognitive symptoms begin to appear in COVID-19 patients, which patient groups are at a higher risk, how long neurological effects may last and what treatments are most effective. In the early stages of understanding COVID-19 and researching the neurological effects, definitive facts around these issues remain unknown.
Our McMaster University laboratory has pioneered the direct measurement of real-time brain function using electroencephalogram-based (EEG-based) neuroimaging to assess neurocognitive function.
We have used EEG-based methods for many years to study the cognitive problems that often accompany brain injuries, including disorders of consciousness such as “vegetative state” (now often called unresponsive wakefulness syndrome), coma and concussion.
Using unique prompts, data collection and machine-learning algorithms, we can now observe brain activity at significantly higher resolution by analyzing results based on data obtained in increments of two to three minutes instead of 20 to 30 minutes. This means we can see rapidly occurring changes in brain function with unprecedented speed. The ability to objectively measure a patient’s brain function has a vital impact on their trajectory, and helps avoid misdiagnosis and provide appropriate treatment.
We’ve even been able to correct some misconceptions about neurocognitive function after a brain injury. In some coma patients, we found rapidly fluctuating brain activity that reflects variations in consciousness — proof of brain activity that had a high risk of being missed by earlier testing methods. We have also identified clear markers of neurocognitive dysfunction in so-called “invisible injuries,” such as mild traumatic brain injuries (concussions), even when the patient’s most recent concussion occurred decades earlier.
Our laboratory partnered with a McMaster neurotech startup called VoxNeuro to make the method available to health-care providers and their patients outside of research settings after requests from our health-care research partners to use the method clinically. Together, we are well-positioned to use these same pioneering assessment methods in COVID-19 patients.
The neurocognitive techniques my colleagues and I have developed use advanced acquisition and analysis methods to examine electrophysiological activity in the brain. The result is electrophysiological recordings and observed responses (called event-related potentials, or ERPs) that have a number of important and in some cases unique features that lend themselves to studying typical cognitive functions like memory, attention and decision making — commonly referred to as mental abilities. They are particularly suitable for testing vulnerable patients.
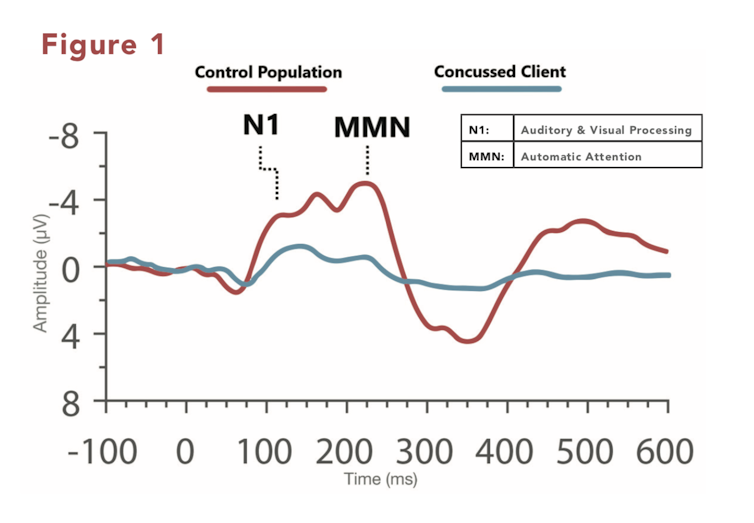
First, recording a person’s electrophysiological responses is non-invasive: no elements of the technology require injections or inhalation of any substances. The technology is compatible with other medical devices such as pacemakers.
Particularly useful for vulnerable patients is its point-of-care feature: we can test a patient at the bedside, in the intensive care unit or in their home. We are able to objectively measure the size of brain responses and the all-important timing of these responses with millisecond accuracy. This reveals specific neurocognitive dysfunctions with great accuracy, which means doctors can tailor treatment and rehabilitation to each patient’s needs.
COVID-19’s “neuro-invasive” features are leading to calls to prepare for the consequences of the daunting relationship between COVID-19 and neurologic pathologies. As more patients recover, we need to continue to monitor them as they return to their everyday lives. Are they functioning as they did before they caught the virus? Or are they experiencing difficulties returning to work, with complications concentrating or maintaining focus?
It’s not enough to monitor a patient until they recover from the obvious life-threatening complications of the illness. Patients require continued monitoring to detect potential long-term effects and treat them proactively. If cognitive consequences go untreated, they can manifest into much worse complications that are harder to fix the longer they are left alone.
There are lessons to be learned from the history of related coronaviruses, where research has noted neuro-invasive features leading to learning and memory problems in adults and children.
Another vital question for health-care response efforts is: How are medical interventions for COVID-19 affecting brain function? Do certain pharmaceutical drugs or ventilators have a negative impact? As much as we need to better understand the virus with objective data, we need to understand, objectively, what interventions help or harm patients at scale. These are the questions that we are looking to answer in upcoming research through engagements with critical care specialists and CanCOVID — Canada’s expert-led COVID-19 response.
It’s only been half a year since this virus started to come into view, and scientists and clinicians are still striving to understand how it works and what damage it leaves behind. As we begin to plan for COVID-19’s massive social, medical and economic legacies, our lab is proud to open a window into how it changes our body’s most complex and important system.![]()
John F. Connolly is a professor and the Senator William McMaster Chair in the Cognitive Neuroscience of Language; director of the ARiEAL Research Centre; and co-director of Language, Memory & Brain Laboratories at McMaster University. This article is republished from The Conversation under a Creative Commons license. Read the original article.


